
Stability is when an atom has a full set of eight valence electrons in their outermost shell (called the octet rule). Metals like to lose electrons to achieve stability, and form positive ions in the process.Beauty salon peroxide to show how difference in concentration affects rate of reaction.Ordinarily it is a colorless gas, but vast quantities of it in space absorb starlight, creating spectacular sights such as the Eagle Nebula (seen by the Hubble Space telescope).įound in: water, sugar, ammonia, rocket fuel, stars & nebulae, and air (External Sample)By weight, 75 percent of the visible universe is hydrogen. Polonium - rare radioactive element, named after Poland (discoverers Marie & Pierre Curie's native country)Ĭhlorine - table salt, disinfectant, bleaching of paper product or clothingīromine - dye, disinfectant, photographic chemicals Tellurium - semiconductors, ceramics, tinting glass Selenium - solar cells, light meters, photocopiers Sulfur - food preservative, rubber product, bleaching & refrigeration Phosphorus - highly reactive/corrosive solid, also found in human body, used in match heads (very flammable)Īntimony - hardens & strengthens lead, semiconductors, batteriesīismuth - carrier for uranium fuel in nuclear reactors, fire extinguishing systems, cosmetics, medicine Nitrogen (largest component of air), builds proteins in cells, DNA/RNA Tin lines steel food cans, mixed with copper makes bronze, Lead resists corrosion - used in ceramics, plumbing, glassmaking Graphite in pencils and powder lubricant, silicon comprises sand and used in semiconductors (computer chips), Thallium is poisonous - not many uses, but sometimes mixed with other compounds to form types of glass. Indium also has a low melting point - used in alloys in thermometers and flat-screen TV's Gallium is solid at room temp but melts in your hand - used in electronic devices Elements with atomic number greater than 92 are manufactured in laboratories and are highly unstable (radioactive)īoron makes boric acid (mild antiseptic), borax (laundry water softener & ant killer) and a small component of silly putty - produces green flame when burnedĪluminum (most abundant metal in Earth's crust) is soft and light - found in baseball bats, drink cans, bikes & cooking utensils.Radium is radioactive (too many protons or too few neutrons causes unstable atom where particles are released) and used in cancer chemotherapy. Calcium in limestone & marble, essential for strong teeth & bones, Strontium gives fireworks their red color, barium in ceramics and some types of glass (and GI x-rays). Sodium chloride is table salt, lithium in batteries, Cesium in clocks, potassium in fireworks, liquid detergents, fertilizers and vitamins, rubidium in photocells (motion detectors)īeryllium in high-speed aircraft, missiles, spacecraft & satellites, Magnesium combined with other metals to form strong but lightweight alloys. 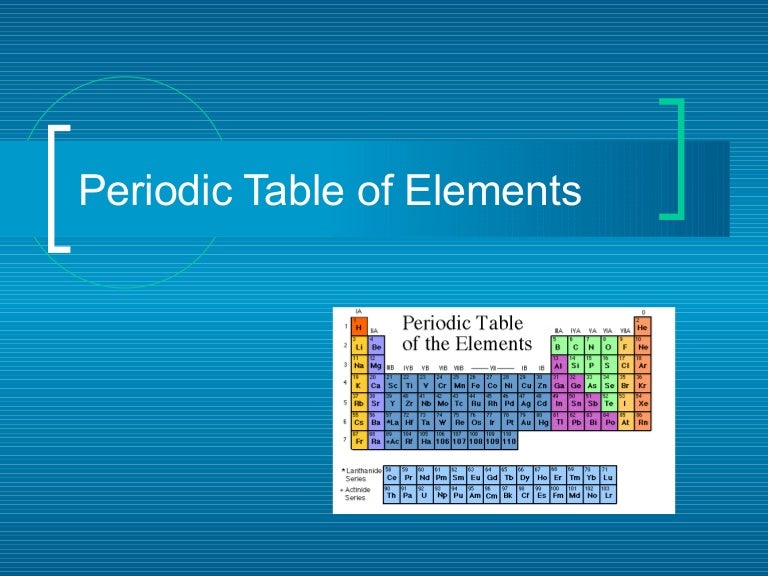 
Patterns: atomic # increases as you move left to rightĮnergy levels increase as you move down a group Like the days of the month, the chemical elements can be arranged in a way that shows a repeating, or PERIODIC pattern.All because of the atoms that form them -ĭifferent atoms have different properties that react depending on their number of protons, electrons & neutrons.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
